Tritium Self Luminous Exit Sign | Red, Green, Black | 2-3 Week Lead Time
Base Code: SL-Tritium


Additional Pendant Mount Kits Click Here
NOTE: Tritium Exit Signs are custom made to order, are non-returnable and must be shipped to a business address.
The Exit Light Company has been a continuous provider of tritium exit signs since 2003 and is a go-to source of these units to the US Government.
An efficient and secure exit sign that uses no electricity or batteries, requires no wires or maintenance and is explosion and weatherproof. Mounting brackets included to allow both top or lateral installation.
Self-luminous Tritium Exit Signs are registered with the U.S. Nuclear Regulatory Center as required by law. Before we can process this order for shipment, you will need to provide End User Information (where the signs will actually be installed).
Disposal:
Need to get rid of expired or damaged Tritium Exit Signs? These signs need to be disposed of safely according to the NRC regulations (failure to do so can result in significant fines). Select the disposal option above if buying replacements. For additional disposals (with no replacements ordered) see our Tritium Disposal service.
Tritium Self-luminous Signs Frequently Asked Questions (FAQs)
How Does a Tritium exit sign work?
Self-luminous signs use the electron from the tritium to provide illumination without the need for a source of electrical power. The process is very similar to that in your television set picture tube where an electron is used to illuminate the front screen of the tube. The electron from tritium however has only a fraction of the energy of the electron in a color TV picture tube. That is why self-luminous signs are not visible in daylight while TV pictures are. Actually, the electron from the tritium has such a low energy that it cannot even penetrate an ordinary sheet of paper. To illuminate, the tritium gas is contained within a hermetically sealed glass tube. The inside surfaces of the tube are coated with a phosphor just like the inside surface of a television picture tube. Electrons emitted by the tritium bombard the phosphor causing it to produce illumination.
How do you turn it on?
You do not have to do anything to activate it. Your sign is always on; just take it into a dark room and turn off the lights to view its illumination.
Why isn't it brighter?
It doesn't need to be. You will not see the tritium lamps illuminating when the normal lighting is on in a building, but the UL required contrast ratio of the face colors makes the exit sign very visible. When you really need it, when the power has gone out, the sign becomes very visible. The brightness of this unit is more than twice the Underwriters Laboratories (UL) minimum requirement.
Is this sign approved?
Yes! It meets the requirements of the National Fire Protection Association Life Safety Code 101 which is the bible for most AHJ's. Other approvals/acceptances include: Underwriters Laboratories, OSHA, NSI, BOCA, ICBO, SBCCI and the FAA (Commercial airliners have tritium exits to protect their passengers)
What is Tritium?
Tritium gas is an isotope of the chemical element hydrogen that contains one proton, two neutrons and is naturally present in the atmosphere. Tritium is an unstable isotope, meaning that its molecular structure is subject to decay (hence the 10 or 20 year lifetime). Unstable isotopes are referred to as radioactive isotopes. In radioactive isotopes, the nucleus, or center, decays to form a different nucleus and a nuclear particle. The nucleus in tritium decays by emitting an electron called a beta particle. The rate at which a radioactive element loses its radioactivity (decays) determines its half-life, the time it takes the element to decay to half its original activity level. Tritium has a half life of approximately 12 1/2 years which is very short compared with many isotopes you may have read about in articles on current events or in high school or college science courses.
Is there any radiation from these signs?
No. Tritium emits a beta electron which cannot even penetrate a piece of paper. The tubes in signs which contain the tritium are mounted inside a high-impact plastic case designed to be tamper and vandal resistant. A clear high-impact plastic shield across the face of the sign provides additional protection and serves as another barrier against accidental damage.
What if a tube breaks?
Should the protective shield and case be penetrated and a tube breaks, releasing the tritium gas, there is no hazard. Because it is hydrogen and therefore lighter than air, when released, the tritium gas is dispersed rapidly and harmlessly into the atmosphere to join the naturally occurring tritium already dispersed. In the highly improbable event that all of the multiple tubes should fracture, the effect is still less than half of that received from naturally occurring radioactive sources during a year, and is similar to the difference between living at sea level and moving to an elevation of 5,000 feet.

UL Listed
This item has been tested and meets safety standards imposed by the Underwriters Laboratories (UL).

Radioluminescent
Self-luminous signs use the electron from the tritium to provide illumination without the need for a source of electrical power.
Features
- UL 924, OSHA and NFPA 101 Life Safety Code, USNRC, ISO 9001
- Listed for wet location
- *** Call for availability of RUSH product ***
- OPERATING TEMPERATURE: -76°F TO +212°F
- Left, right, or no arrow configuration
- Explosion-proof, weatherproof
- Hard plastic housing with red face, no maintenance
- Easy installation, no wires
- Concealed mounting
- Suitable for floor proximity mounting
- 10 or 20 year lifetime
- 100 Ft Viewing Distance
- NRC Regulated product, nonreturnable, non-refundable
- Single or double-sided option
Previous Product Codes: SL-Tritium
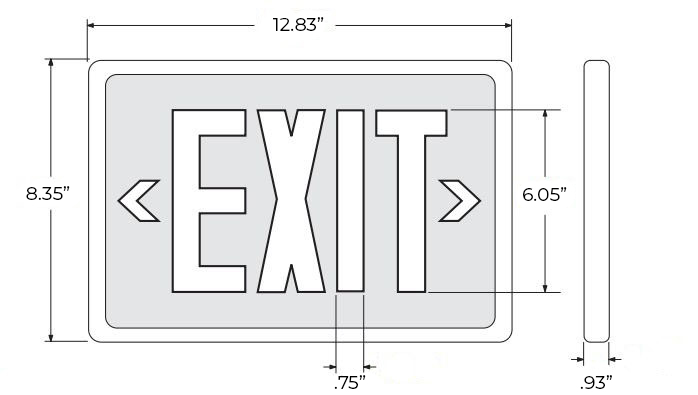
Specifications
| Dimensions (May Vary by Model) | |
| Width | 12.83" |
| Height | 8.35" |
| Depth | 0.93" |
| Weight | 3.00 lb |
What's included?
- Tritium Exit Sign
- One "EXIT" Faceplate (with optional arrows)
- One Blank Faceplate
- Mounting Canopy, Installation Instructions & Hardware
Construction
The SL Tritium series is an explosion proof and weatherproof Tritium powerless Exit Sign encased with a high impact, fire-retardant ABS plastic.
Illumination
The SL Tritium Exit Sign does not illuminate when normal lighting is on in the building. When the power goes out, the sign illuminates bright emergency lighting, more than twice what the Underwriters Laboratories (UL) requires.
Emergency Operation
The SL Tritium exit sign series are always on. Depending on the sign lifetime selected, these signs will operate for 10 or 20 years.
Electrical Components
Tritium Exit Signs contain no electrical components. They feature Betalight tubes manufactured from borosilicate (hard) glass internally coated with zinc sulfide phosphor and filled with tritium tubes.
Installation
The SL series installs in minutes. It is suitable for wall mount, ceiling mount, pendant mount*, and recessed mount*. 2 color matching arrow covers are included to cover up desired directional arrow.
*Pendant and Recessed Mount sold separately
![]() Mounting Configurations Explained
Mounting Configurations Explained
![]() View Installation Instructions
View Installation Instructions
Dimensions
View Common Questions or Ask Your Own Below




